Supplying the NHS – a year on, what has the pandemic taught us?
Attain have considerable experience in shaping, managing, and supporting NHS Trusts and CCG commissioners to undertake procurements and advise on commercial considerations and supply chain implications across the health sector.
Rod Skinner and Jonathan Edwards, Senior Managers within the Procurement Community at Attain, provide some insights into Supply Chain Resilience and how they relate to Procurement and Commissioning.
The last 12 months have seen unparalleled times within the NHS for procurement and supply chain functions, however, for both procurement and supply chain design, if approached correctly, they can anticipate and mitigate against supply continuity risks for critical items, such as medicines, PPE and key equipment. In this article we lay out some strategic considerations.
NHS procurement and supply chain functions need to focus on ‘resilience’ to support future pandemic responses and take a more system wide approach with contingencies at both national at regional level. NHS leaders must recognise resilience a key component of supply chain solutions and move away from a ‘cost only’ focus on awarding contracts.
Supply chain resilience
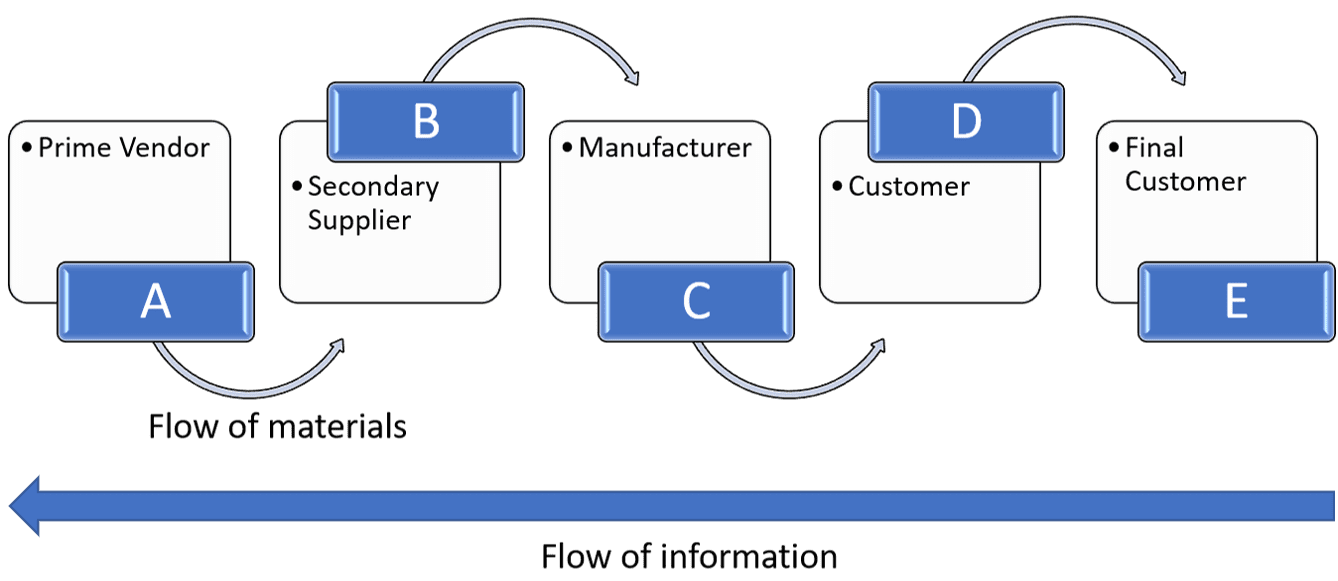
A supply chain can be considered as the flow of materials and information from end to end in ‘originator to consumption’ with the flow of materials moving typically from the stages of A) a prime vendor to B) a secondary supplier, to C) a manufacturer, to D) a customer and finally E) final customer with information flowing back through the supply chain.
‘Supply chain resilience’ we consider as the ability to quickly recover from supply interruptions in order to fulfil customer requirements. Specifically, within an NHS setting, we see the following as being key supply chain areas of critical importance and consideration for NHS leaders in terms of building necessary resilience:
- Clinical Requirements – establishing the true ‘need’ – through regional Trusts; creation of clinical reference groups considering product and service specifications and certifications, agreeing staff training or product fitting needs and approval for alternative contingency products removing variation between Trusts and supporting procurement teams
- Demand Planning – NHS demand planners to re-calculate key material/service requirements before procurement teams begin any sourcing activities
- Supply Planning & Product Sourcing – Professional Category Management approach, key/essential product lines and multiple sources identified (UK, Europe and Global)
During the initial peak of the COVID-19 outbreak it became apparent of an NHS overreliance on supply chains associated with ‘just in time’ lean, but long, lead times associated with vulnerable international supplier capacity due to variables, such as, unavailability of labour, materials and freight limitations. The NHS thus scrambled for all the vital products it could get whilst immediately using up any form of safety stock built up.
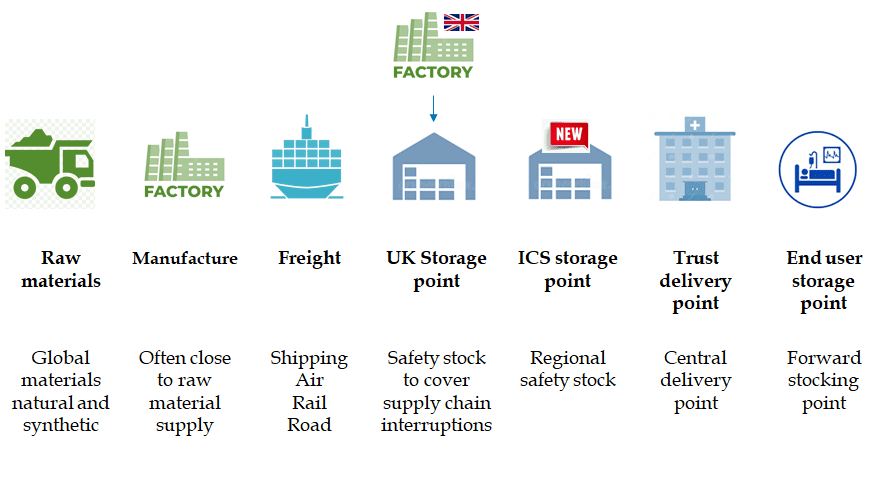
We propose an alternative approach; that primary and secondary supply routes be established to mitigate supply disruption. Diversifying sourcing bases with a multi-sourcing approach can include continual global sourcing but also adopting a nearshoring approach, building resilience by reducing lead time and delivery risk using supply routes closer to or within the UK.
Key Considerations:
- Regional and Trust level contingency stocks – The additional capacity from multiple sourcing can supplement UK storage point safety stock from which we propose an additional storage point at Regional (Integrated Care System) level which Trusts can add extra resilience on a local level.
- Stock transparency across storerooms and site – End-to-end value chain visibility is critical. Key stock held to be visible across the Trust(s) ensuring it can be moved to where it is most needed. Regional stock visibility is important for key products enabling stock to be repositioned to where it is most needed in case of a surge in demand supporting any regional ‘mutual aid’ initiatives.
- Managing consumption of product – In an emergency such as the response to Covid it is important that the amount of stock used in each Trust area is visible, enabling a forward view of when stock will be exhausted. This informs the procurement team of what additional stock needs to be sourced identifying any differences in clinical practice or stock leakage and reducing waste.
- Clinical standardisation – For contingency products we propose a regional based product list that have been tested and accepted be compiled and the establishment of a regional group to provide a rapid approval or rejection of products that procurement teams propose to source creating opportunities to place higher volumes among multiple suppliers in turn enhancing further supply chain resilience. Regional standardisation also improves safety and removes the need for additional clinical training as staff work across sites.
- Stress testing supply chains – understanding required time for points in the supply chain to be restored following disruption and the maximum duration that supply chains can match supply with demand after a particular supply facility disruption. Including dependence on out of region suppliers, concentration of suppliers, depth and interconnectivity of the supply chain and inventory volatility.
Summary
Supply chain resilience within the NHS needs to become business as usual beginning with optimising stock creating regional storage facility’s to manage contingency stock together with consideration for ‘reusable’ stock for critical lines (e.g. laundering gowns, decontaminating masks) and also the development of systems to ensure stock and consumption visibility across the supply chain.
From a demand and sourcing perspective development of a multi-sourcing approach to diversify sourcing base together with increased understanding of demand with focus on how to create harmonisation / variation reduction between Trusts bringing together infection control and clinical leads to agree product standards for contingency products with one central point of product review.
Finally, the need to stress test the supply chain in order to gauge resilience to its ability to quickly recover from supply interruptions in order to fulfil customer requirements.
If you would like more details about this or how we can support you please email at contacts@Attain.co.uk
More about our expertise
Contact us to discuss your challenges
Get in touch with us to learn more about how we can support you in addressing your challenges.
Complete our simple form and we’ll call you right back, alternatively just email: